After reading hundreds of articles and research papers, we started to come across several papers and theories that seemed like the pieces of a puzzle whose image was hidden. After several years and much effort, the pieces finally started fitting together to reveal a compelling picture.
 The following is our interpretation of what may be behind the increase in autoimmune and food-related disorders:
The following is our interpretation of what may be behind the increase in autoimmune and food-related disorders:
Though it is a complicated, poorly understood, and controversial condition, we believe that leaky gut syndrome is highly relevant and may play a crucial role in the development of celiac disease, Crohn’s disease, ulcerative colitis, rheumatoid arthritis, asthma, chronic fatigue syndrome, psoriasis and much more.
We understand that leaky gut syndrome is a widely debated condition and that not all health professionals believe it to be related to (or a direct cause of) disease. As research progresses, many doctors, dietitians, and researchers are becoming aware of the condition and seek ways to test for and treat it.
If you haven’t heard of leaky gut syndrome, please read below:
Leaky gut syndrome is the easier-to-say term for increased intestinal wall permeability. It just means that gut wall is easier to cross than it should be. The gut wall begins to be more porous and develop holes. Basically, your gut is leaking things it shouldn’t into your bloodstream.
Leaky gut is thought to be caused or worsened by certain components of foods (more on that later), cytotoxic drugs, NSAIDS (non-steroidal anti-inflammatory drugs), irradiation of food, antibiotics, unbalanced gut flora, excessive alcohol consumption and compromised immunity.
The intestinal lining is on the front lines of our immune system. We like to think of it as castle wall – we let the drawbridge down for visitors we know (like food and resource deliveries) but we leave it up to keep out invaders.
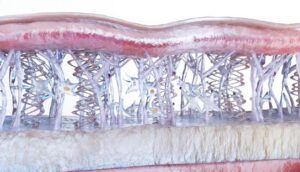 The other layers of this sophisticated defense is called the epithelium. A single layer of epithelial cells normally stay connected together by tight junctions. These tight junctions are how the passage of nutrients is regulated in the digestive tract. The epithelial cells in the gut are tipped with finger-like projections called villi. When food is digested, villi absorb the nutrients and transport them through the epithelial cell into the bloodstream.
The other layers of this sophisticated defense is called the epithelium. A single layer of epithelial cells normally stay connected together by tight junctions. These tight junctions are how the passage of nutrients is regulated in the digestive tract. The epithelial cells in the gut are tipped with finger-like projections called villi. When food is digested, villi absorb the nutrients and transport them through the epithelial cell into the bloodstream.
When the digestive process is functioning normally, the tight junctions stay closed and only nutrients are allowed to pass through into the blood stream. When something goes wrong, the tight junctions become permeable or “open” and allow un-screened molecules through the border and into the bloodstream. Examples of things that can get through in this situation are bacteria, pathogens, yeast, incompletely digested food, lectins and more. That’s why we call it a leaky gut.
As this process continues over time, the intestinal lining can become damaged and even leakier, allowing even more “undesirables” through the intestinal wall and directly into the bloodstream.
Typically, this increase in offenders in the blood will make the liver and kidneys work that much harder to filter it all out. As the gut becomes increasingly damaged, the liver or kidneys may not be able to keep up with the constant flow of bacteria, pathogens, yeast, undigested macro-nutrients and waste products escaping through the gut lining.
As more invaders get through, overworking and overwhelming liver and kidneys, they are able to wreak havoc systemically (throughout the body).
When these offenders attach to the cells lining the gut, an immune response is triggered which can lead to collateral damage of healthy cells. This can lead to another chain of events in which the immune system begins to recognize certain molecules from food as invaders, calls for an immune response whenever you eat those foods, which can then cause even more collateral damage. This collateral damage can be experienced as bloating, cramps, diarrhea, inflammation, joint pain, skin rashes, headaches, malabsorption and more.
As the result of continuous immune response and corresponding collateral damage, the gut becomes more and more damaged. If you are frequently eating foods that it recognizes as an invader – it has no time to heal. Healthy cells are destroyed and those microvilli we talked about earlier are not able to do their job, which prevents your body from getting all the nutrients that you need, which in turn leads to all sorts of problems such as a weakened immune system or nutritional deficiencies.
To make matters worse, as your immune systems weakens, you become more susceptible to illness from the stream of junk (toxins, bacteria, pathogens, etc.) flowing through your leaky gut.
 If this vicious cycle continues for weeks or months or years, you body may ultimately end up fighting itself, potentially leading to the initiation of autoimmune diseases such as as Crohn’s disease, ulcerative colitis, multiple sclerosis, type 1 diabetes, lupus, rheumatoid arthritis, chronic fatigue syndrome, fibromyalgia, vasculitis, urticaria (hives), alopecia areata, polymyalgia rheumatica, Raynaud’s syndrome, vitiligo, thyroiditis, and Sjogren’s syndrome.
If this vicious cycle continues for weeks or months or years, you body may ultimately end up fighting itself, potentially leading to the initiation of autoimmune diseases such as as Crohn’s disease, ulcerative colitis, multiple sclerosis, type 1 diabetes, lupus, rheumatoid arthritis, chronic fatigue syndrome, fibromyalgia, vasculitis, urticaria (hives), alopecia areata, polymyalgia rheumatica, Raynaud’s syndrome, vitiligo, thyroiditis, and Sjogren’s syndrome.
With me so far? Great!
Let’s go deeper and talk about the probable triggers that start off this awful process.
Some of the most interesting emerging research regarding digestion and autoimmune disease has to do with a few factors that could be triggers for opening the draw bridge (tight junctions) of the gut. A few key factors that appear to be the likely causes of leaky gut are zonulin, gluten (the gliadin portion), and some lectins.
Zonulin is a protein that modulated the permeability of the tight junctions in the gut. So far, it is the only “key” that we know the human body produces. Zonulin upregulation has been implicated in the pathogenesis of several autoimmune diseases including celiac and type 1 diabetes. Zonulin is currently being studied as a potential target for celiac treatment.
Gliadin (1/2 of the protein complex we commonly refer to as gluten) has been shown to active (upregulate) zonulin signaling in everyone, regardless of celiac status, leading to the opening of tight junctions in the gut and leading to increased gut permeability. Gliadin is found in wheat, barley, rye, and triticale, which are all grains found nearly everywhere in our modern food supply. There are three main types of gliadin (α, ϒ, and ω), and they all produce an immune response in those with celiac disease.
 The term lectin refers to a specific class of proteins that bind to carbohydrate moieties. They are found in almost all plants and animals but the variety that we are most interested in are concentrated in certain plants and some dairy products. These lectins bind to glycoproteins and glycolipids (sugar-coated proteins and fats) found on the surface of human and other animal cells. This binding allows for agglutination (clumping) and sometimes can produce an immune response. They can cause agglutination of blood cells and they can bind to the cells that line the small intestine. Nasty business.
The term lectin refers to a specific class of proteins that bind to carbohydrate moieties. They are found in almost all plants and animals but the variety that we are most interested in are concentrated in certain plants and some dairy products. These lectins bind to glycoproteins and glycolipids (sugar-coated proteins and fats) found on the surface of human and other animal cells. This binding allows for agglutination (clumping) and sometimes can produce an immune response. They can cause agglutination of blood cells and they can bind to the cells that line the small intestine. Nasty business.
The most interesting research I’ve seen in this area came from Alessio Fasano, M.D. and his team. Dr. Fasano is a world-renowned pediatric gastroenterologist, the W. Allan Walker Chair of Pediatrics at Harvard Medical School, Vice Chair of Basic, Translational, and Clinical Research and Division Chief of Pediatric Gastroenterology and Nutrition at the MassGeneral Hospital for Children in Boston and really fun to watch lecture.
He proposes that altering or upregulating (as gluten does) zonulin pathways can cause autoimmune and inflammatory disorders. More interestingly, he thinks that these diseases can be all but reversed by reestablishing the zonulin-dependent tight junctions of the intestine.
Here’s the Abstract from Dr. Fasano’s paper:
The primary functions of the gastrointestinal tract have traditionally been perceived to be limited to the digestion and absorption of nutrients and to electrolytes and water homeostasis. A more attentive analysis of the anatomic and functional arrangement of the gastrointestinal tract, however, suggests that another extremely important function of this organ is its ability to regulate the trafficking of macromolecules between the environment and the host through a barrier mechanism. Together with the gut-associated lymphoid tissue and the neuroendocrine network, the intestinal epithelial barrier, with its intercellular tight junctions, controls the equilibrium between tolerance and immunity to non-self antigens. Zonulin is the only physiological modulator of intercellular tight junctions described so far that is involved in trafficking of macromolecules and, therefore, in tolerance/immune response balance. When the finely tuned zonulin pathway is deregulated in genetically susceptible individuals, both intestinal and extraintestinal autoimmune, inflammatory, and neoplastic disorders can occur. This new paradigm subverts traditional theories underlying the development of these diseases and suggests that these processes can be arrested if the interplay between genes and environmental triggers is prevented by reestablishing the zonulin-dependent intestinal barrier function. This review is timely given the increased interest in the role of a “leaky gut” in the pathogenesis of several pathological conditions targeting both the intestine and extraintestinal organs.
So zonulin is the “key” that opens the tight junctions of the intestine and when the finely tuned zonulin pathway is disrupted, autoimmune, inflammatory, and neoplastic (tumor-related) diseases can occur. The worst part is that once the body mounts a defense against any particular protein that escapes, it becomes trained to react to those proteins every time they appear which can lead to chronic inflammation.
So far, the only triggers that we have found for the zounlin pathways are certain gut bacteria in the small intestine and gluten. Dr. Fasano’s research indicates that gliadin increases zonulin levels in everyone, regardless of celiac status. As zonulin levels rise, the tight junctions of the gut get less and less…tight. This allows bacteria, pathogens, and macromolecules of undigested food to pass directly into the bloodstream.
In conclusion, Dr. Fasano states:
The classical paradigm of inflammatory pathogenesis involving specific genetic makeup and exposure to environmental triggers has been challenged recently by the addition of a third element, the loss of intestinal barrier function. Genetic predisposition, miscommunication between innate and adaptive immunity, exposure to environmental triggers, and loss of intestinal barrier function secondary to the activation of the zonulin pathway by food-derived environmental triggers or changes in gut microbiota all seem to be key ingredients involved in the pathogenesis of inflammation, autoimmunity, and cancer. This new theory implies that once the pathological process is activated, it is not auto-perpetuating. Rather, it can be modulated or even reversed by preventing the continuous interplay between genes and the environment. Since zonulin-dependent TJ dysfunction allows such interactions, new therapeutic strategies aimed at reestablishing the intestinal barrier function by downregulating the zonulin pathway offer innovative and not-yet-explored approaches for the management of these debilitating chronic diseases.
Dr. Fasano’s team has also found evidence that gluten may be responsible for the pathogenesis of all autoimmune disease (not just celiac disease) due to the upregulation of zonulin and the subsequent loosing of tight junctions. It also suggests that this process plays a role in inflammatory diseases such as Chron’s and rheumatoid arthritis.
Check out his piece in Scientific American or read the abstract below:
OBJECTIVE:
Little is known about the interaction of gliadin with intestinal epithelial cells and the mechanism(s) through which gliadin crosses the intestinal epithelial barrier. We investigated whether gliadin has any immediate effect on zonulin release and signaling.
MATERIAL AND METHODS:
Both ex vivo human small intestines and intestinal cell monolayers were exposed to gliadin, and zonulin release and changes in paracellular permeability were monitored in the presence and absence of zonulin antagonism. Zonulin binding, cytoskeletal rearrangement, and zonula occludens-1 (ZO-1) redistribution were evaluated by immunofluorescence microscopy. Tight junction occludin and ZO-1 gene expression was evaluated by real-time polymerase chain reaction (PCR).
RESULTS:
When exposed to gliadin, zonulin receptor-positive IEC6 and Caco2 cells released zonulin in the cell medium with subsequent zonulin binding to the cell surface, rearrangement of the cell cytoskeleton, loss of occludin-ZO1 protein-protein interaction, and increased monolayer permeability. Pretreatment with the zonulin antagonist FZI/0 blocked these changes without affecting zonulin release. When exposed to luminal gliadin, intestinal biopsies from celiac patients in remission expressed a sustained luminal zonulin release and increase in intestinal permeability that was blocked by FZI/0 pretreatment. Conversely, biopsies from non-celiac patients demonstrated a limited, transient zonulin release which was paralleled by an increase in intestinal permeability that never reached the level of permeability seen in celiac disease (CD) tissues. Chronic gliadin exposure caused down-regulation of both ZO-1 and occludin gene expression.
CONCLUSIONS:
Based on our results, we concluded that gliadin activates zonulin signaling irrespective of the genetic expression of autoimmunity, leading to increased intestinal permeability to macromolecules.
To summarize, this research indicates that gluten (gliadin) causes an enormous release of zonulin. Zonulin opens the tight junctions of the intestinal epithelial cells and creates the opportunity for systemic inflammation and autoimmune response. Most interestingly, this process does not appear to happen only in those with celiac disease. Gluten is a danger to everyone.
All right, that covers gluten and zonulin pretty well. Let’s talk the sneakiest of the bunch: lectins. As I said before, lectins are a class of proteins that bind onto the carbohydrates in our cell walls.
Lectins are present in about 30-40% of the American diet but they are especially concentrated in grains (wheat is the worst), seeds, nuts, legumes (soy is the worst), nightshade plants (potatoes, tomatoes, eggplant, peppers, etc), and dairy. Importantly, lectins are not fully degraded by heat or by digestion, which means they often reach the small intestine more or less intact.
Some ingested lectins will make it through the gut wall by a process called endocytosis, which may allow them access to the blood and lymph system. From here, they may enter the liver, pancreas or other organs. Experts believe that about 5 percent of ingested lectins will enter the bloodstream and, depending on the individuals specific glycoconjugates (what type of sugar is bound to their cell walls), bind to various tissues in the body such as nervous tissue or connective tissue or even the bladder.
Some lectins that we consume in everyday foods can bind to the sugars in the cell walls of the gut or in the blood. This can cause an immune response, leading to inflammation, intestinal damage, altered gut flora, malabsorption, decreased cellular repair, cellular death, and eventually disease.
Now, that’s in HEALTHY GUT. Let’s just think about how much worse it can be in a leaky gut. In a leaky gut situation, more complete lectins can flow directly into the bloodstream and bind to tissues throughout the body potentially causing autoimmune mayhem. As discussed earlier, this could lead to chronic inflammation or disease.
The effect of lectins on those with an established autoimmune disease such as Crohn’s or celiac could be even worse. The epithelial cells of the small intestine are renewing at a faster rate and immature cells are more glycosylated than mature cells making them more susceptible to lectin binding.
Though the area is still being researched, studies have shown that lectins are linked to many autoimmune disorders like Crohn’s, ulcerative colitis, arthritis, celiac disease, and more. The correlation between intestinal lining damage, gut permeability, and chronic autoimmune response is very compelling.
For example, in wheat, gliadin, a component of gluten and an iso-lectin of wheat germ agglutinin (WGA), is capable of activating NF kappa beta proteins which, when up-regulated, are involved in almost every acute and chronic inflammatory disorder including neurodegenerative disease, inflammatory bowel disease, infectious and autoimmune diseases.
 There is also good evidence that lectins like WGA can initiate allergic reactions in the gut causing the release of IL-4, IL-13, and histamine from human basophils producing noticeable allergic symptoms. Additionally, WGA has been shown to interfere with protein digestion and increase gut permeability.
There is also good evidence that lectins like WGA can initiate allergic reactions in the gut causing the release of IL-4, IL-13, and histamine from human basophils producing noticeable allergic symptoms. Additionally, WGA has been shown to interfere with protein digestion and increase gut permeability.
Lectins found in peanuts, kidney beans, and soybeans are all capable of attaching to various bodily tissues and having deleterious effects. Of course, it is not as simple as that. Our individual genotype (the genes we have) and phenotype (how those genes are expressed) play a role in how lectins affect any of us. Almost everyone tested so far have antibodies to some dietary lectins in their bloodstream meaning that their immune systems have mounted a defense against those lectins at some point. In fact, many allergists agree that most of what we call food allergies are actually immune responses to the lectins in the foods we eat.
Now, it’s almost impossible to avoid eating lectins because they are in just about every plant and animal to varying degrees (even if you try to avoid them you’ll still end up eating small amounts given their omnipresence in nature). However, lectins are definitely more concentrated in some sources than others.
I said it earlier but it is worth repeating; Here is a list of foods with a high concentration of lectins: All grains (wheat is the worst offender), dairy, nuts, legumes (soy is the worst offender), and nightshade plants (potatoes, tomatoes, eggplant, tamarios, tomatillos, pepinos, pimentos, paprika, cayenne, and peppers of all kinds except black pepper).
All of this evidence leads to the conclusion that it is probable that all types of autoimmune diseases, many inflammatory diseases, and possibly neoplastic diseases share a common thread: leaky gut syndrome. That is not to say that leaky gut syndrome is causing these disease, but it does seem to be a necessary condition for them to occur.
Here is a scenario that demonstrates how one can go from a healthy state to a disease state after eating certain foods:
1. First, let’s suppose that you are genetically predisposed (which means you were probably “born with it”) to suffer from certain (or multiple) diseases which are activated by diet.
2. You eat gluten or other foods containing wheat and or lectins (legumes, nightshade vegetables, etc.).
3. You are exposed to a variety of environmental toxins in addition to the naturally occurring compounds found in what you eat.
4. Over time, that exposure causes leaky gut syndrome.
5. Once the gut becomes “leaky”, toxins, bacteria, pathogens, lectins, incompletely digested nutrients, and waste products flow from the gut directly into the bloodstream.
6. This can wreak havoc on the systems of the body (affecting different people in different ways.)
7. The immune system scrambles to deal with these “invaders.”
8. Immune-response damages your own cells in the process of attacking the invaders.
9. Because the invaders are attached to your own cells, the body begins to recognize these cells as bad guys and attacks healthy cells. (That’s why it is called “autoimmune.”)
10. This process is repeated over the course of weeks or years eventually leading to the pathogenesis of an autoimmune disease, chronic inflammatory disease, or neoplastic disorder.
(Please look at our references for this article to gain a deeper understanding of these concepts and theories)
1. Fasano, A. Leaky gut and autoimmune diseases. Clinical Reviews in Allergy & Immunology, Feb;42(1):71-8. doi: 10.1007/s12016-011-8291-x.
2. Fasano, A. Zonulin and Its Regulation of Intestinal Barrier Function: The Biological Door to Inflammation, Autoimmunity, and Cancer. Physiol Rev January 1, 2011 vol. 91 no. 1 151-175
3. Fasano, A. Gliadin, zonulin and gut permeability: Effects on celiac and non-celiac and non-celiac intestinal mucosa and intestinal cell lines. Scandinavian Journal of Gastroenterology, 2006; 41: 408 Á/419
4. Pusztai A, Ewen SW, Grant G, Brown DS, Stewart JC, Peumans WJ, Van Damme EJ, Bardocz S. Antinutritive effects of wheat germ agglutinin and other N-acetylglucosamine-specific lectins. Br J Nutr. 1993 Jul;70(1):313-321.
5. Jones, David S., ed.. Textbook of Functional Medicine. Gig Harbor:The Institute for Functional Medicine, 2005, 303.
6. Watzl B, Neudecker C, Hansch GM, Rechkemmer G, Pool-Zobel BL. Dietary wheat germ agglutinin modulates ovalbumin-induced immune responses in Brown Norway rats. Br J Nutr. 2001 Apr;85(4):483-90.
7. Eur. J. Immunology. 1999. Mar;29(3):918-27.
8. Falth-Magnusson K., et al. Elevated levels of serum antibodies to the lectin wheat germ agglutinin in celiac children lend support to the gluten-lectin theory of celiac disease. Pediatr Allergy Immunol. May 1995; 6(2): 98-102.
9. Hollander D, Vadheim CM, Brettholz E, Pertersen GM, Delahunty T, Rotter JI. Increased intestinal permeability in patients with Crohn’s disease and their relatives. A possible etiologic factor. Ann Intern Med, December 1986; 105(6):883-85.
10. Gut 1999. May; 44(5):709-14
11. J Cell Physiol. 2001 Feb;186(2):282-287.
12. Pusztai A. Dietary lectins are metabolic signals for the gut and modulate immune and hormonal functions. Eur J Clin Nutr. 1993 Oct; 47(10):691-699 ( Pusztai A Rowett Research Institute, Bucksburn, Aberdeen, UK.
13. High Frequency of Extractable Nuclear Autoantibodies in Wheat-Related DisordersYuanyuan Yang,1 Karthik Krishna,2 Payal Deshpande,1 Vinodh Ranganathan,2 Vasanth Jayaraman,2Tianhao Wang,2 Kang Bei,2 and Hari Krishnamurthy2

Etiam magna arcu, ullamcorper ut pulvinar et, ornare sit amet ligula. Aliquam vitae bibendum lorem. Cras id dui lectus.
63739 street lorem ipsum City, Country
+12 (0) 345 678 9
info@company.com

Take the first step towards a happier gut. Get 5% OFF your next purchase of DigestShield.